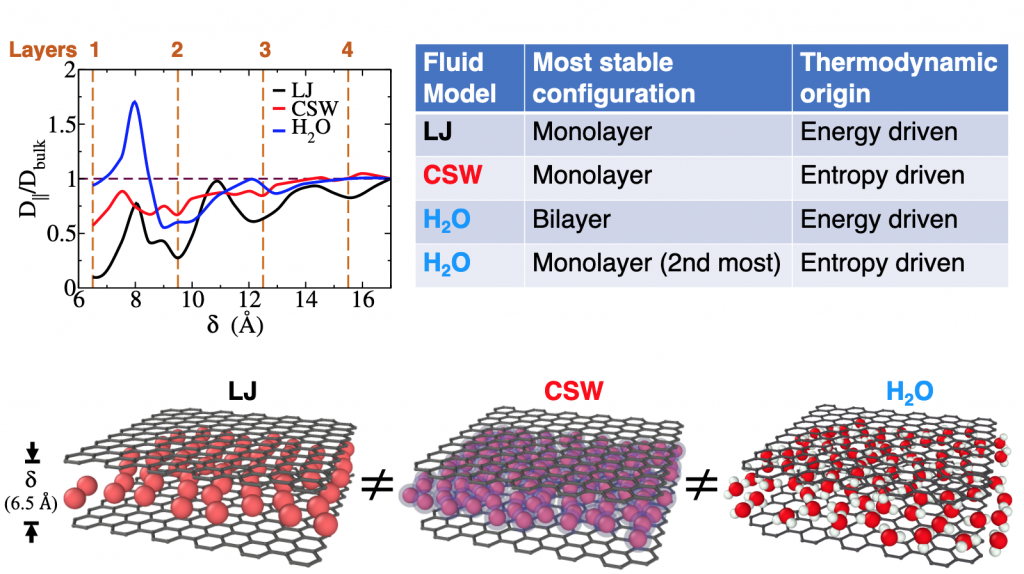
Living organisms, viruses, and technological devices, have water layers between their cells or parts and can die or stop working when dehydrated. But why water and not any other fluid? What makes water unique when it is in these tiny pores? In their publication on ACS Nano Dr. Fabio Leoni of the Department of Physics of the University La Sapienza of Rome, and Dr. Carles Calero and Dr. Giancarlo Franzese of the Department of Condensed Matter Physics of the University of Barcelona and group leader at Statistical Physics of Bio-Nano Systems and Complex Matter from the Institute of Nanoscience and Nanotechnology at UB (IN2UB) explain why the water in a pore tinier than one-millionth of a hair moves faster than free water, while other fluids do not. It all depends on the peculiar hydrogen bond interaction of water and it might be a key factor contributing to the solution of one of the Sustainable Development Goals defined by the United Nations Department of Economic and Social Affairs, the one about clean water and sanitation. Indeed, nanometer-size graphene pores are one of the most investigated membranes for water sanitation and treatment. Furthermore, the result of this joint project between the UB Physics School and Natural Science School at La Sapienza relates to the switching behavior observed in a hydrated graphene nano-memristor, that is a nanotechnology devices where the electric charge rules the magnetic flux, when the confined water changes from one to more layers (Ref).
Nanoconfined Fluids: Uniqueness of Water Compared to Other Liquids. Fabio Leoni, Carles Calero, and Giancarlo Franzese. ACS Nano, November 22, 2021