Ouldouz Ghashghaei, Marina Pedrola, Francesca Seghetti, Victor V. Martin, Ricardo Zavarce, Michal Babiak, Jiri Novacek, Frederick Hartung, Katharina M. Rolfes, Thomas Haarmann-Stemmann, Rodolfo Lavilla
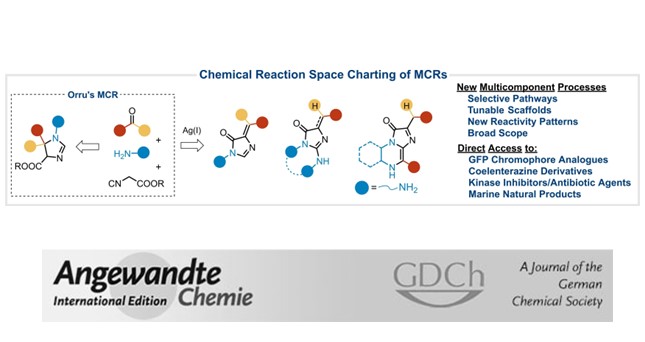
By engaging indole aldehydes in an isocyanide multicomponent reaction, the polarity inversion of the indole moiety along the transformation triggers an extension of the domino process, leading to a variety of unprecedented fused, linked, and bridged scaffolds. The processes can be run in parallel and the adducts display remarkable bioactivity as potent ligands of the aryl hydrocarbon receptor.
https://onlinelibrary.wiley.com/doi/abs/10.1002/ange.202011253