ER (ESTROGEN RECEPTOR)
Estrogen receptors (ERs) have a high affinity for estrogens and act by regulating gene expression through various mechanisms: in a classical way, regulating transcription after binding to estrogen response elements in DNA, or in a non-classical way, interacting with other transcription factors such as AP-1 or Sp-1.
Finally, they can signal outside the nucleus, activating signaling cascades that include MAPKs, Src, and Akt. Activation of these signaling pathways can end in the regulation of transcription or in the non-transcriptional regulation of enzymes, ion channels or other proteins involved in cell function.
Mammals have two estrogen receptor subtypes: ER alpha (ERα, also known as ESR1) and ER beta (ERβ or ESR2). There is a third type of estrogen receptor, called the “G-protein coupled estrogen receptor” (GPER), which structure is that of a G-protein coupled receptor.
Ligands
The main ligands of ERs are estrogens (steroid hormones), although ERα has a higher affinity for estrone and ERβ for estriol. However, it should be noted that they are promiscuous receptors that bind exogenous compounds, called environmental estrogens, that often act as endocrine disruptors.
Classification
Estrogen receptors (ER) are members of the NR3 subfamily of the large nuclear receptor (NR) superfamily.
In turn, based on their affinity for the ligand and its mode of action, they are classified as “endocrine” and Type I receptors.
Structure and interactions
All members of the nuclear receptor family share a general multi-domain structure. ERs are made up of the following six domains:
- A/B (NTD / AF-1): Coactivator binding domain with DNA transactivation function (AF-1), in N-terminal position. Important for ligand independent transcriptional regulation.
- C (DBD): highly conserved DNA binding region, containing two “zinc fingers” that interact with specific hormonal response elements (HRE motifs, in this case called ERE), and mediating receptor dimerization.
- D: Junction “hinge region” of the C and E domains, which contains a nuclear localization signal and gives flexibility to the receptor.
- E/F (LBD + H12 / AF-2): Ligand-binding domain, in C-terminal position, and with transcriptional activation function (AF-2) by binding to coactivators in Helix 12 (H12). Important for ligand-dependent transcriptional regulation.
Helix 12 is a highly conserved region within LBD. When it binds to the ligand, its structural configuration is modified, resulting in an active form (bound to the agonist) or inactive form (bound to the antagonist) in the transcription regulation.
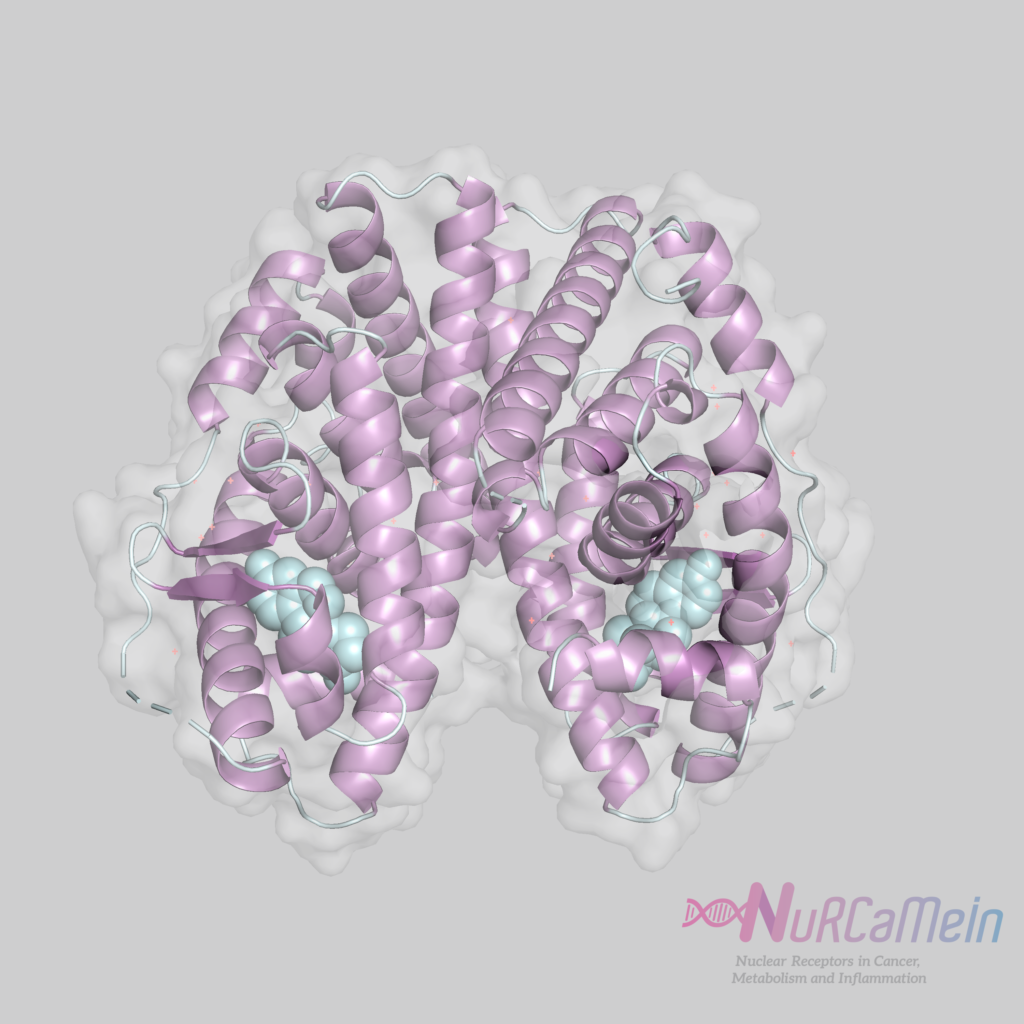
ER structure
As described above, ERs act in the following ways:
- Classic nuclear mechanism: by binding to estrogens, ERs form homo or heterodimers that bind to the ERE motif within the promoter of a target gene, thereby regulating its transcription.
- Non-classical nuclear mechanism: ERs can be indirectly linked to the AP-1 DNA motif to regulate, for example, transcription through the FOS / JUN dimer (anchoring mechanism).
- Mechanism initiated outside the nucleus: ERs can bind to the membrane after palmitoylation, where they activate signaling cascades that may or may not end in the regulation of transcription.
- Ligand independent mechanism: growth factors (IGF, EGF) can phosphorylate ERs through growth factor membrane receptor (GFR) mediated intracellular signaling pathways. Thus ERs can regulate gene expression despite the absence of ligand.
Expression
ERαs are expressed mainly in the mammary and pituitary glands, uterus, ovaries (thecal cells), bones, male reproductive organs (testes and epididymis), prostate (stroma), liver and adipose tissue.
While ERβs do it mainly in the prostate (epithelium), ovaries (granulosa cells), lung, colon, adipose tissue and immune system.
Both ER subtypes are remarkably expressed in the cardiovascular and central nervous systems.
Main functions
ERs regulate key physiological functions in various body systems, such as the reproductive system (function and development of the ovaries), skeletal, cardiovascular, and central nervous systems; as well as in specific tissues such as the breast, prostate and ovary.
ERαs have a more prominent role in the mammary gland and the uterus, and in the preservation of skeletal homeostasis and the regulation of metabolism.
ERβs appear to have increased activity in the central nervous system and immune system, and generally counteracts the cellular hyperproliferation promoted by ERαs in tissues such as the breast and uterus.
Both receptors have important functions in the glucidic and lipidic metabolism, being expressed in adipocytes, skeletal muscle, liver and endocrine pancreas.
ERs in the NuRCaMeIn Network
- Instituto de Investigación Desarrollo e Innovación en Biotecnología Sanitaria de Elche (IDiBE) | Universidad Miguel Hernández de Elche
-
Estrogen Receptors and Diabetes | Nurcamein
- Cancer Heterogeneity Lab | CICbioGUNE
- Nuclear Receptors and Breast Cancer | Nurcamein
References
- Estrogen Receptors: New Directions in the New Millennium, Sylvia C. Hewitt and Kenneth S Korach et al. | Endocrine Reviews
- Estrogen Hormone Biology, Katherine J. Hamilton et al. | Current Topics in Developmental Biology
- Estrogen, Estrogen Receptor and Lung Cancer, Li-Han Hsu et al. | International Journal of Molecular Science
- ncer, Li-Han Hsu et al. | International Journal of Molecular Sciences
