Signal transduction, cell cycle and cancer (Signaling and checkpoints of cell cycle). PI: Neus Agell Jané
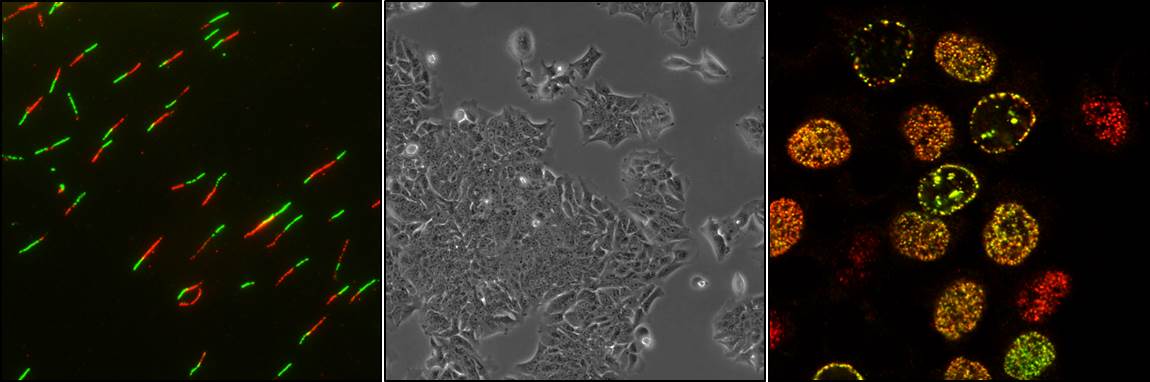
The general objective of the group is to investigate the signal transduction pathways that regulate the progression of the cell cycle and its control mechanisms (checkpoints), as well as to analyze the implication of their alterations in oncogenesis. In relation to this general objective, in recent years we have developed the following lines of research:
- New strategies to inhibit oncogenic KRAS.
- Replicative stress: response of untransformed cells versus tumor cells.
The group is also part of the IDIBAPS Research Institute within the "Intracellular Compartments and Signaling" research team in the Oncology and Hematology area:
http://www.idibaps.org/recerca/309/compartiments-intracel%C2%B7lulars-i-senyalitzacio
KEYWORDS: Colorectal cancer; pancreatic cancer; RAS; KRAS phosphorylation; gene expression; cell polarization; tumorigenesis; calmodulin; small molecule inhibitors; peptidomimetics; DNA replication; replication stress; replication fork stability; hydroxyurea.

Dra. Neus Agell Jané
Full Professor of Cell Biology
neusagell@ub.edu
Dra. Montserrat Jaumot Pijoan
Associate Professor
mjaumot@ub.edu

Dra. Sònia Brun Lozano
Higher Technique in Cell Biology
soniabrun@ub.edu
Baraa Abuasaker
Pre-doctoral student, UB Pre-doc Scholarship
babuasaker@ub.edu
Jesús Daniel Gómez Zepeda
Pre-doctoral student, UB Pre-doc Scholarship
jgomezze7@alumnes.ub.edu

Fernando Unzueta Payeras
Pre-doctoral student, FPU Scholarship (MINECO)
f.unzueta@ub.edu

Alejandro Pérez Venteo
Master's student in Biomedicine
aperezve84@alumnes.ub.edu
Manuel Fernández Nogueira
Degree student in Biology Sciences
mfernano51@alumnes.ub.edu
- New strategies to inhibit oncogenic KRAS
Ras proteins are small GTPases that regulate signal transduction pathways that control cell proliferation, differentiation, survival, and apoptosis. Oncogenic mutations of the KRAS isoform are very common in colorectal, pancreatic, and lung cancers. Unfortunately, no good inhibitors of KRAS function have yet been found that can be used in therapy. In order to deepen the knowledge of the regulation of KRAS and search for strategies to inhibit its oncogenic action, our group: on the one hand, investigates the role of KRAS phosphorylation at serine 181; and on the other, it identifies proteins that interact with KRAS and regulate its function, until now not described. Summarizing, we have shown that the phosphorylation of KRAS regulates its oncogenic activity, and that calmodulin and hnRNPA2 proteins specifically bind to KRAS. Moreover, calmodulin inhibits the phosphorylation of KRAS at serine 181, and thus modulates the functionality of the non-oncogenic and oncogenic forms; and hnRNPA2 interacts better with the phosphorylated form of KRAS and favours its signalling in adenocarcinoma cells of the pancreas.
- Replicative stress: response of untransformed cells versus tumour cells
Genomic instability is a hallmark of tumour cells, and DNA replication is the most vulnerable cell cycle event to genomic instability. Replicative stress plays an important role in oncogenesis, but at the same time it could become a good target in cancer therapy. That is why our group investigates the responses to replicative stress of tumour cells versus non-transformed cells, whether it is induced by nucleotide depletion, DNA damage, or by overexpression of an oncogene such as KRAS. Our results demonstrate that after severe replicative stress, the non-transformed cells go into senescence while most tumour cells are able to recover and continue to proliferate but acquiring alterations in the genome.
A peptidemimetic inhibidor of RAS as targeted therapy for pancreatic cancer. Prova de concepte 2021
Fundacio Bosch i Gimpera
F2I-PdCl-2021-003
PI: Neus Agell
2022
Peptidomiméticos inhibidores de KRAS como terapia dirigida para el cáncer de páncreas. Pruebas de concepto 2021
Ministerio de Ciencia e Innovación
PDC2021-121119-I00
PI: Neus Agell
01/12/2021 - 30/11/2023
Nuevas interacciones de KRAS relevantes para su actividad oncogénica y efecto del estrés de replicación en la actividad transcripcional
Ministerio de Ciencia e Innovación
PID2019-105483RB-I00
PI: Neus Agell
01/01/2020 - 01/7/2023
La fosforilación de K-Ras, su interacción con calmodulina, y la respuesta al estrés replicativo, en la progresión del cáncer colorectal y en el diseño de nuevas terapias. SAF2016-76239-R
Ministerio de Economia y Competitividad
SAF2016-76239-R
PI: Neus Agell
30/12/2016 - 29/12/2019
For more information for PI publications click in the link:
ORCID: https://orcid.org/0000-0002-1205-6074
Baraa Abuasaker, Eduardo Garrido, Marta Vilaplana, Jesús Daniel Gómez-Zepeda, Sonia Brun, Marta Garcia-Cajide, Caroline Mauvezin, Montserrat Jaumot, Maria Dolors Pujol, Jaime Rubio-Martínez, Neus Agell. α4-α5 Helices on Surface of KRAS Can Accommodate Small Compounds That Increase KRAS Signaling While Inducing CRC Cell Death. Int J Mol Sci . 2023 Jan 1;24(1): 748. doi: 10.3390/ ijms24010748. PMID: 36614192
Chiara Pallara, Debora Cabot, Josep Rivas, Sonia Brun, Jesús Seco, Baraa Abuasaker, Teresa Tarragó, Montserrat Jaumot, Roger Prades, Neus Agell. Peptidomimetics designed to bind to RAS effector domain are promising cancer therapeutic compounds. Sci Rep. 2022 Sep 22;12(1): 15810. doi: 10.1038/ s41598-022-19703-6. PMID: 36138080
Sonia Feu, Fernando Unzueta, Amaia Ercilla, Alejandro Pérez-Venteo, Montserrat Jaumot, Neus Agell. RAD51 is a druggable target that sustains replication fork progression upon DNA replication stress. PLoS One. 2022 Aug 15; 17(8): e0266645. doi: 10.1371/ journal.pone. 0266645. eCollection 2022. PMID: 35969531
Cabot D, Brun S, Paco N, Ginesta MM, ……. Agell N. KRAS phosphorylation regulates cell polarization and tumorigenic properties in colorectal cancer. Oncogene. 2021 Sep; 40(38): 5730-5740. doi: 10.1038/ s41388-021-01967-3. Epub 2021 Jul 31. PMID: 34333552
Pelletier J, Riaño-Canalias F, Almacellas E, Mauvezin C, Samino S, Feu S, Menoyo S, Domostegui A, Garcia-Cajide M, Salazar R, Cortés C, Marcos R, Tauler A, Yanes O, Agell N, Kozma SC, Gentilella A, Thomas G. Nucleotide depletion reveals the impaired ribosome biogenesis checkpoint as a barrier against DNA damage. EMBO J. 2020 Jul 1;39 (13): e103838. doi: 10.15252/ embj. 2019103838. Epub 2020 Jun 2. PMID: 32484960 Free PMC article.
Tebar F, Chavero A, Agell N, Lu A, Rentero C, Enrich C, Grewal T. Pleiotropic Roles of Calmodulin in the Regulation of KRas and Rac1 GTPases: Functional Diversity in Health and Disease. Int J Mol Sci. 2020 May 23;21 (10): 3680. doi: 10.3390/ ijms21103680. PMID: 32456244 Free PMC article. Review.
Feu, S.; Unzueta, F.; Llopis, A.; Semple, J.; Ercilla, A.; Guaita-Esteruelas, S.; Jaumot, M.; Freire, R.; Agell N. OZF is a Claspin-interacting protein essential to maintain the replication fork progression rate under replication stress. 900388 - The FASEB Journal. 34 - 5, pp. 6907 - 6919. (Estats Units d’Amèrica): The Federation of American Society of Experimental Biology, 2020. ISSN 0892-6638. DOI: https:// doi.org/ 10.1096/ fj.201901926R.
Ercilla, A.; Feu, S.; Aranda, S.; Llopis, A.; Brynjólfsdóttir, S.H.; Sørensen, C.S.; Toledo, L.I.; Agell, N. Acute hydroxyurea-induced replication blockade results in replisome components disengagement from nascent DNA without causing fork collapse. 909449 - Cellular and Molecular Life Sciences. (Suïssa): Springer Verlag, 2019. ISSN 1420-682X. DOI: https: //doi.org/ 10.1007/ s00018-019-03206-1. Handle: http://hdl.handle.net/2445/137967. PMID: 31297568.
Garrido, Eduardo; Jaumot, Montserrat; Agell, Neus; Granadino-Roldan, Jose M.; Rubio-Martinez, Jaime. Toward understanding calmodulin plasticity by molecular dynamics. 915755 - Future Medicinal Chemistry. 11 - 9, pp. 975 -991. (Regne Unit): Future Science, 2019. ISSN 1756-8919. DOI: https: //doi.org/ 10.4155/ fmc-2018-0323. PMID: 31140879.
Garrido E, Lázaro J, Jaumot M, Agell N, Rubio-Martinez J. Modeling and subtleties of K-Ras and Calmodulin interaction. PLoS Comput Biol. 2018 Oct 30; 14(10): e1006552. doi: 10.1371/ journal.pcbi.1006552. PMID: 30376570
Wangsa D, Quintanilla I, Torabi K, Vila-Casadesús M, Ercilla A, Klus G, Yuce Z, Galofré C, Cuatrecasas M, Lozano JJ, Agell N, Cimini D, Castells A, Ried T, Camps J. FASEB J. Near-tetraploid cancer cells show chromosome instability triggered by replication stress and exhibit enhanced invasiveness. 2018 Jul; 32(7): 3502-3517. doi: 10. 1096/ fj.201700247RR. PMID: 29452566
Brun S, Abella N, Berciano MT, Tapia O, Jaumot M, Freire R, Lafarga M, Agell N. SUMO regulates p21Cip1 intracellular distribution and with p21Cip1 facilitates multiprotein complex formation in the nucleolus upon DNA damage. PLoS One. 2017 Jun 5; 12(6): e0178925. doi: 10.1371/ journal.pone.0178925. PMID: 28582471
Ercilla A, Llopis A, Feu S, Aranda S, Ernfors P, Freire R, Agell N. New origin firing is inhibited by APC/CCdh1 activation in S-phase after severe replication stress. Nucleic Acids Res. 2016 Jun 2; 44(10): 4745-62. doi: 10.1093/ nar/ gkw132. Epub 2016 Mar 2. PMID: 26939887
Domínguez D, Feijoo P, Bernal A, Ercilla A, Agell N, Genescà A, Tusell L. Centrosome aberrations in human mammary epithelial cells driven by cooperative interactions between p16INK4a deficiency and telomere-dependent genotoxic stress. Oncotarget. 2015 Sep 29; 6(29): 28238-56. doi: 10.18632/ oncotarget.4958. PMID: 26318587
Barceló C, Etchin J, Mansour MR, Sanda T, Ginesta MM, Sanchez-Arévalo Lobo VJ, Real FX, Capellà G, Estanyol JM, Jaumot M, Look AT, Agell N. Ribonucleoprotein HNRNPA2B1 interacts with and regulates oncogenic KRAS in pancreatic ductal adenocarcinoma cells. Gastroenterology. 2014 Oct; 147(4): 882-892.e8. doi: 10.1053/ j.gastro. 2014.06.041. PMID: 24998203
Barceló C, Paco N, Morell M, Alvarez-Moya B, Bota-Rabassedas N, Jaumot M, Vilardell F, Capella G, Agell N. Phosphorylation at Ser-181 of oncogenic KRAS is required for tumor growth. Cancer Res. 2014 Feb 15; 74(4): 1190-9. doi: 10.1158/ 0008-5472. CAN-13-1750. PMID: 24371225
- Offers from the University of Barcelona:
- Offers from the Research Group:
At the moment, there are no job offers from this Research Group